Early results of the ACO experiment: directionally right, but impact is still small
In this study, the authors compare metrics for Medicare beneficiaries assigned to the 32 ACOs part of the Pioneer program vs. matched beneficiaries who were not in an ACO. With respect to costs, they find that compared to contemporaneous trends observed in non-ACO members, the ACO beneficiaries yearly spending was approximately $100 below trend (a 1% savings). In a hint of a reversal of a secular trend in health care, office spending visit expenditure increased more in ACOs, while it was the reverse for hospital-based services which was the driver of the overall decrease in cost. Quality measures were relatively the same, except for preventive services for diabetes which trended better in ACOs. Looking at sub-groups of ACOs, those that had the best relative improvement in performance were those that started out with high spending showing the potential of the program to improve inefficient systems, but also highlighting the challenge in creating incentives that will attract currently above average performers. Performance Differences in Year 1 of Pioneer Accountable Care Organizations (free access)
Continuing progress in cystic fibrosis
Three years ago, ivacaftor (Kalydeco – Vertex), was approved as the first drug the defect that leads to cystic fibrosis (CF), but unfortunately, it was only shown to work in a specific mutation that is a rare cause of CF. Since then further work has focused on helping those with a more common mutation (Phe500del, 45% of CF patients) and in this article, the results of two phase 3 studies of ivacaftor and lumacaftor (Orkambi, also from Vertex) show a modest improvement of the treatment arms vs. placebo. In particular, patients on the combination therapy had fewer hospitalizations related to CF, and put on weight (a challenge for patients with CF which also affects the digestive tract in addition to the lungs). Barring the unexpected, lumacaftor should be approved by the FDA in the next few weeks. Lumacaftor–Ivacaftor in Patients with Cystic Fibrosis Homozygous for Phe508del CFTR; Another Beginning for Cystic Fibrosis Therapy (free access)
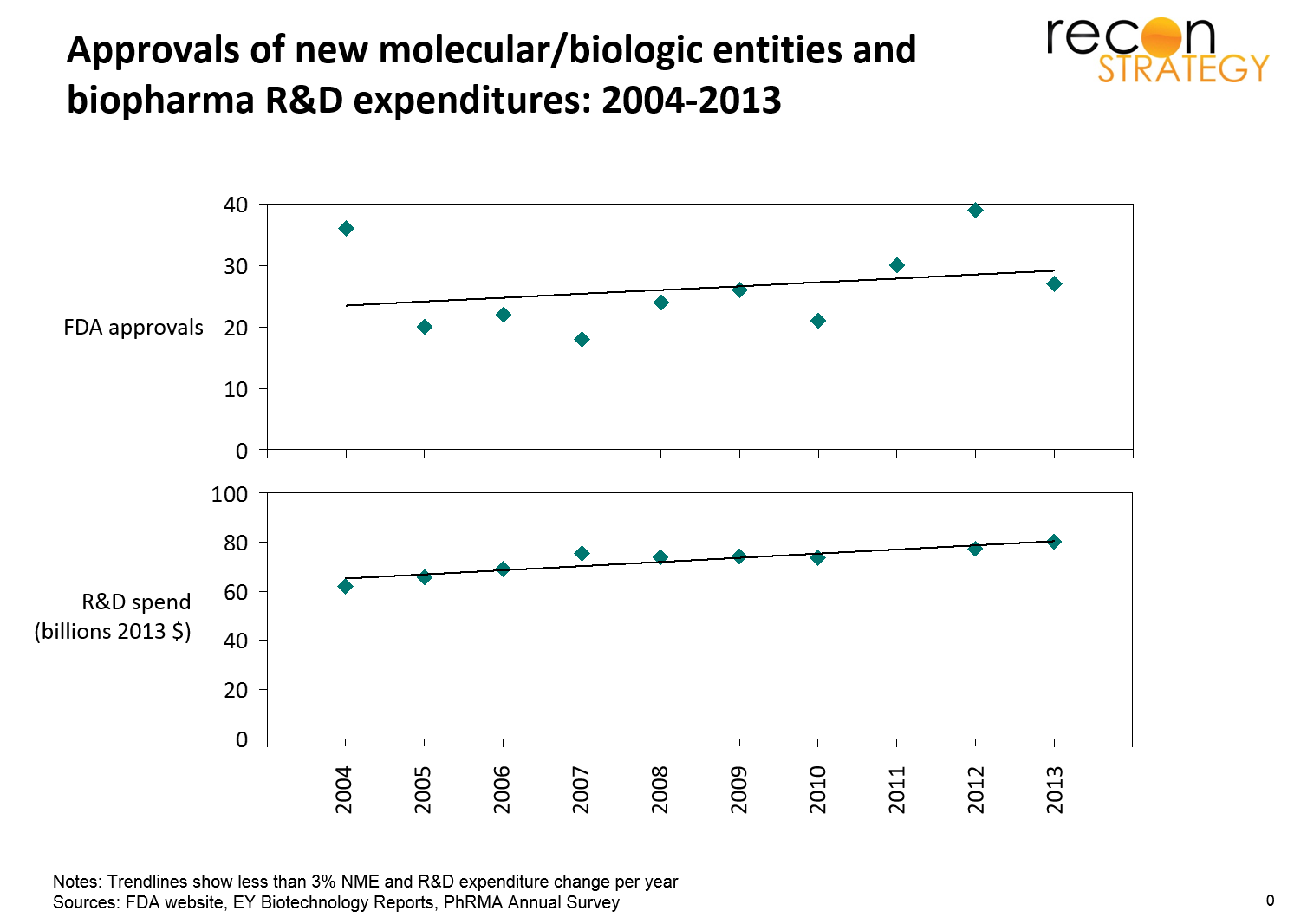
The cost of discovering and developing new drugs
A perspective questioning a recent of estimate by the Tufts Center for the Study of Drug Development that it costs $2.6 billion to develop a drug. A bottom up approach to such estimates is complex (although feasible) because it relies on many parameters – principally, the transition rate from each stage to the next, and the cost and duration linked with each stage. An alternative point of view is top down; i.e. using the yearly Biopharma R&D investment and yearly output in terms of FDA approvals. While the relative timing of investment vs. approval can make the analysis complicated, it turns out that both have been weakly varying over the last decade (see figure below), so that on average, a yearly industry R&D investment of about 72 billion (2013 US dollars) is yielding about 26 new drugs each year – which is very much consistent with the estimate from Tufts. And to reiterate something that cannot be said too many times, the vast majority of the costs come from those drugs that fail and never make it to market, either because of toxicity, or lack of efficacy, or because a better therapy got there first. The $2.6 Billion Pill — Methodologic and Policy Considerations; The Cost of Drug Development (free access)
Conflict of interest in medicine
A series of three thought-provoking papers, Dr. Linda Rosenbaum addresses the thorny issue of relations between the practitioners of medicine and the vendors of medicine (and more specifically Biopharma). In essence, the argument is that the pendulum has swung too far: a culture of purity from industry contact has arisen within medicine that it so extreme that it has created its own conflict of interest. The personal advantage to be seen as a pharmascold (or at least not to cross them) trumps a rational assessment of the trade-offs that collaboration with industry can bring to health. We at Recon also believe that the time is ripe for change and that a shift from a fee for service mentality to a value-based approach to healthcare (this across the board including industry and providers) is likely to help. Reconnecting the Dots — Reinterpreting Industry–Physician Relations; Understanding Bias — The Case for Careful Study; Beyond Moral Outrage — Weighing the Trade-Offs of COI Regulation (free access)
A CVS Caremark-sponsored study on smoking cessation
Not long ago, CVS stopped selling tobacco product and now they seem to have doubled down by sponsoring with the NIH a study on the use of financial incentives to promote smoking cessation. In this work patients were randomized to usual care or one of four interventions where the financial pay-off for quitting was always $800 but structured in different ways – a pure reward, or a reward combined with $150 deposit at risk (therefore a downside of $150 for a failure to quit); individual performance, or group performance (either collaborative where rewards are shared, or competitive where a quitter can gain the deposit of someone who fails to quit). All financial incentives approaches led to significantly higher abstinence rate than usual care, but the differences between group and individual incentives were not significant. The willingness of people to engage in a deposit scheme was much lower than for the pure reward scheme (14% vs. 90%), but when they did engage in the deposit scheme, their rate of abstinence at 6 months was much higher than for those engaged in the reward scheme (52% vs. 17%). And as an interesting side effect, CVS is developing a reputation in population health. Randomized Trial of Four Financial-Incentive Programs for Smoking Cessation (free access)
The New England Journal of Medicine is a premier medical journal covering many topics of interest to the health sector. In this monthly series we offer a brief overview highlights that might be of interest to our clients and others.