PDF: An ADC renaissance: a resurgence of antibody drug conjugates for cancer
Summary:
Antibody-drug conjugates (ADCs) have seen a record number of approvals in 2020. This comes after a first era of intense activity in the early 2000s followed by a period of relative quiescence from the late 2000s to mid-2010s. To understand how the nature of the ADC pipeline has changed over time, we have reviewed all 88 ADC oncology programs that have reached phase 2 development and beyond from 1997 onward. We found that, currently, the main contributor to growth is the development of ADCs which use antibodies directed to targets that had previously been pursued with non-conjugated antibody programs without reaching approval. There may be a broader opportunity to build early into programs “conjugated option(s)” in addition to the base naked antibody option.
A revitalization of the ADC pipeline
Clearly the dominant trend of the cancer landscape in the last few years has been the rise of immuno-oncology (IO), both in the form of explicit asset classes (e.g. with targets such as PD-L1 or CAR-T technology) but also as a new paradigm to more generally understand tumor response to treatment. In the shadow of this sea-change, it would be easy to miss that ADCs approvals have reached a record number this year, with 3 approved in the first 7 months of the year. This resurgence comes after a quiescent period in ADC development, where in the entire decade of 2005-2015 only 3 ADCs were approved. To better understand these trends, we have reviewed all 88 ADC oncology programs that have reached phase 2 development and beyond from 1997 onward in the context of the overall >300 antibody programs that got that far during the same period
Figure 1
As shown in Figure 1, the overall interest in antibody-based oncology drugs has increased over the past two and a half decades, with ever larger numbers entering phase 2 at each time period. ADCs represented almost 50% of all antibody-based drugs entering phase 2 from 2000-2002. However, in the following decade from 2003-2011, development activity with ADCs waned even though overall antibody work kept increasing. This trend reversed in the mid-2010s with the proportion of antibodies that are ADCs increasing again.
The first ADC peak can be attributed to the initial development of antibody-based therapies together with an incomplete understanding of the ability of naked antibodies to cause cell death. As accumulated clinical experience made clear that many naked antibodies can have potent anti-tumor activity, the additional technical burden of drug conjugation became less attractive. As a result, while the number of antibodies in development for cancer shot up, the fraction that were ADCs dropped. Which brings us to the question, why the current revival?
Part of the story certainly lies with technical advances allowing the creation of better ADCs through more reliable linkers, more consistent conjugation, and more potent conjugates (see Beck et al. 2017[i] for a comprehensive review). However, we also wanted to understand how today’s pipeline is different than that of the first golden era of ADCs in the early 2000s. In particular, we had as a preliminary hypothesis that some of the new activity would be driven by life cycle management of an approved antibody threatened by up and coming biosimilars. As we will show, this is a relatively small driver of growth.
Many of today’s ADCs in development are reusing previously researched but unapproved targets
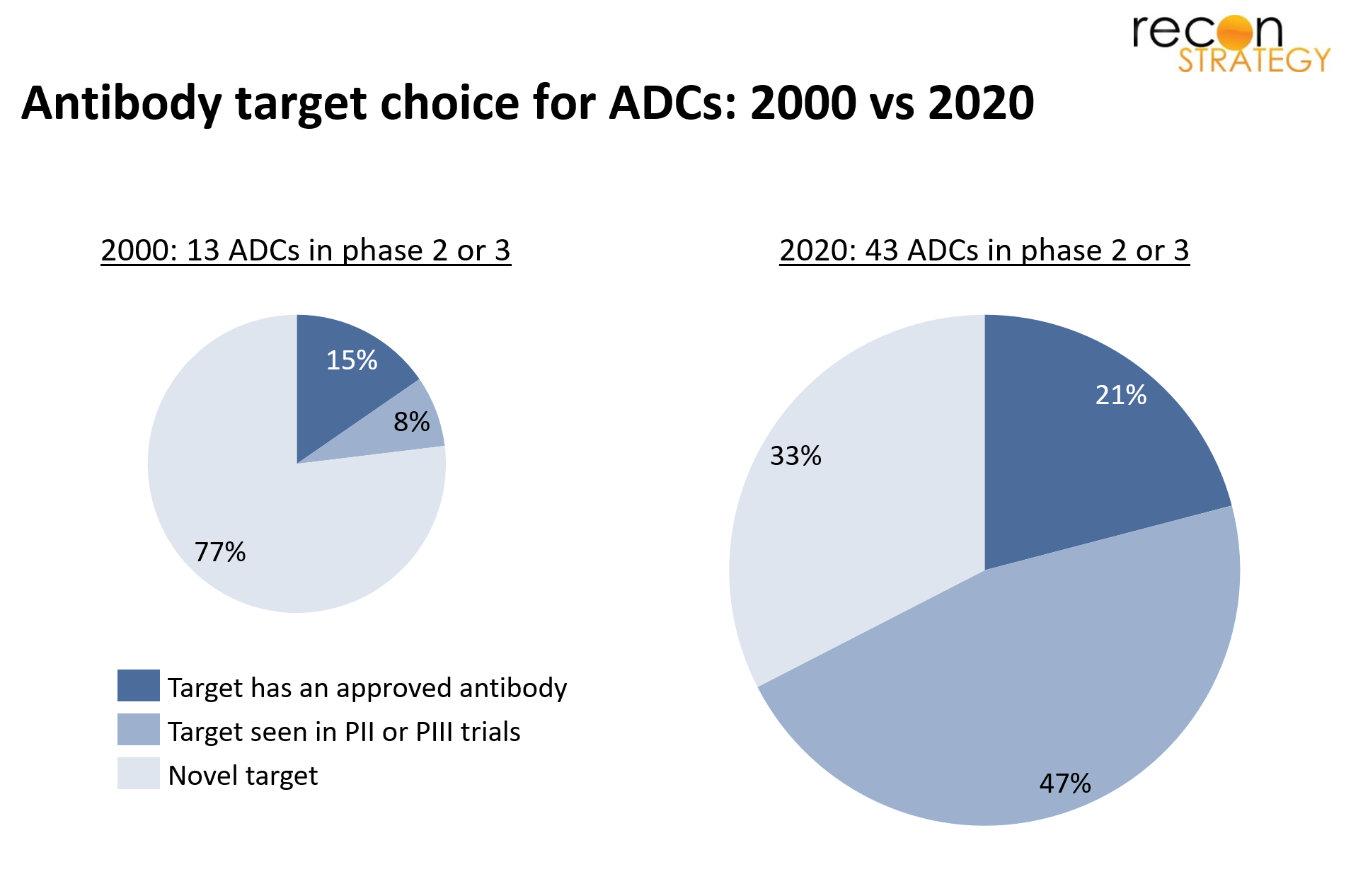
In 2000, during the first peak of ADC activity, 13 ADCs were in late clinical stages (Phase 2 or 3). Unsurprisingly given that the overall approach of using antibodies was still nascent at the time, 77% of those ADCs were aimed at a novel antibody targets which had never reached late clinical development before.
In 2020, during the current ADC renaissance, the nature of the pipeline has changed; 43 ADCs are in late clinical stages, and of those only 33% are aimed at a novel antibody target. The dominant contribution (nearly 47%) is from ADC programs that are reusing targets that were once in development for “naked” antibodies but never approved (Figure 2).
Figure 2
For example, lintuzumab, an antibody directed at CD33, was in development against acute myelogenous leukemia (AML), but failed to show improved efficacy over standard of care in a phase 3 trial. As a result development was discontinued. Fast forward a decade, lintuzumab has been licensed by Actinium Pharmaceuticals who has conjugated it with the radioisotope Ac-225, and it is currently in development for AML.
Another example in solid tumors is patritumab, an antibody directed at HER-3, which was in development against non-small cell lung cancer (NSCLC) but failed to show a benefit vs. placebo in a phase 2 study. Daiichi Sankyo has now conjugated patritumab with a topoisomerase inhibitor and it is currently in development for NSCLC and breast cancer.
In addition to reusing targets for which antibodies went through clinical development but were never approved, a small group of ADCs in the current pipeline are using targets for which there exist approved antibodies. The archetype of this class is trastuzumab which Roche conjugated with emtansine to create Kadcyla (approved in 2013). Since then, a number of other companies have either created outright biosimilars to Kadcyla, or coupled trastuzumab with a different cytotoxic than emtansine, or engineered a different antibody than trastuzumab against HER2 which they then conjugate to a cytotoxic moiety.
Three potential implications to consider:
First, the existence of a large number of programs that are pursuing targets for which naked antibodies have not had success, most of the time many years prior, hints at the possibility that for many antibody programs, an ADC version should be considered as a back-up from the get-go. This would be much like what is common in small molecule programs with a back-up compound waiting in the wings with a 1-2 year lag should the lead asset stumble.
Second, and conversely to the previous point, antibodies that have reached development but never made is to approval are legion. As we have shown, some are now being potentially salvaged through the addition of a conjugate, but there may be more that could be exploited through this approach.
Finally, in an industry that is notorious for its “all-or-nothing” outcomes and where failure often represents the near complete annihilation of large investments, ADCs are an example of an alternative paradigm. Because components of an ADC can be developed and then mixed and matched with existing technology, there is a possibility to hybridize both ‘me-too’ drugs and completely novel drug development, and allow for a more incremental drug development process. In the long run, one could envision a modularized process whereby a library of “targeting agents”, “effector agents”, and “linker agents”, each with their own characterized properties would be available to create customized drug candidates with a higher degree of predictability than a new product created ab initio.
[i] Beck, A., Goetsch, L., Dumontet, C., & Corvaïa, N. (2017). Strategies and challenges for the next generation of antibody–drug conjugates. Nature reviews Drug discovery, 16(5), 315-337.