New molecular diagnostics, especially tests based on next-generation sequencing and gene-panels, could revolutionize how diseases are evaluated. But experience shows that insurers won’t cover these tests just because they’re available – they need to improve clinical care. Here, we explore how researchers and test developers can prove value, and improve their chances at broad clinical uptake.
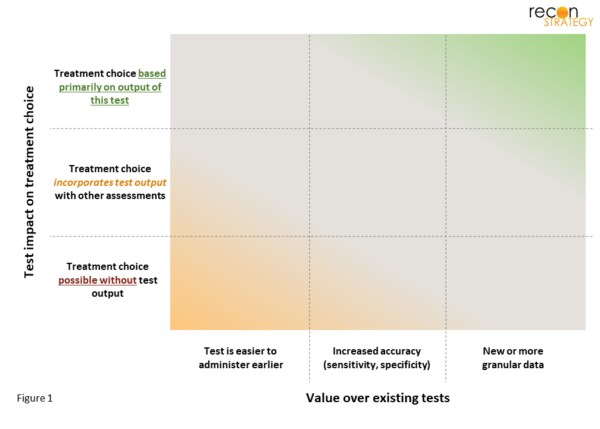
Outside the niche realm of self-pay (where buyers have very different expectations[1]), the business opportunity of new molecular tests is driven by payer-coverage and reimbursement. Payers are beginning to manage[2] high-cost (>$1,000) lab tests just as carefully as high-cost (>$10,000) drugs; they want to see data that shows the impact of the test on patient-outcomes. How tests can change outcomes hinges on two key elements:
- Does the test result play a significant role in treatment decisions?
- Does the test provide information that leads to different treatment choices than other evaluations? This could be due to:
- A unique type of data, or
- Greater sensitivity or specificity of the same data, or
- A test that has lower barriers to use (easier / lower risk).
Using these factors as a framework (Figure 1), it becomes clear which tests will readily gain uptake (upper-right corner) vs. those that may struggle to gain coverage (bottom-left).
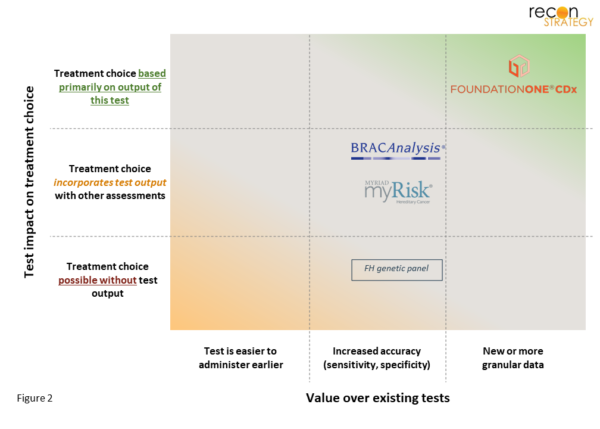
Established tests highlight the importance of how tests inform treatment choice.
A few examples of long-marketed tests illustrate how this framework separates tests with high coverage from those with low coverage (Figure 2).
Tests for targetable cancer-mutations directly inform which immuno/chemotherapy to use.
Today, comprehensive genetic panels (CGPs) such as FoundationOne®, are broadly covered for all cancer patients. These tests detect the most common (and targetable) mutations in tumors, which clinicians use to select optimal treatment (it’s actionable). While the same data can be found in traditional chemical stains of tumor samples, these detect just 1-2 mutations per analysis. In contrast, CGPs simultaneously test for dozens of potential mutations, which eliminates the risk that a relevant mutation is missed due to limited tissue samples (thereby lowering the chance a key mutation is missed and not treated).
Tests for mutations that drastically increase cancer-risk inform preventative care planning.
For some individuals, inherited genes (germline) lead to a much higher (>3x) risk of cancer. Some of these cancers can be prevented by prophylactic surgery or detected earlier with increased surveillance – examples include breast and ovarian cancers. The risk for these cancers can be evaluated by numerous tests, including BRACAnalysis® and myRisk®. However, while carrier status is actionable; it does not always affect outcome – up to 25% of carriers [3] never develop cancer at all, presumably even fewer before changing insurer. As a result, the impact on outcomes (from an insurer’s perspective) is uncertain. As a result, payers typically limit these tests to those with known risk-factors (e.g., at least two-close family members or specific ethnicities).[4]
There is limited value in identifying the genetic drivers of a disease if treatments stay the same.
This test-type typically has tight restrictions on coverage. As an example, consider familial hypercholesterolemia – a condition that increases the risk of coronary artery disease (and major cardiac events) 20-fold.[5] The disease is usually detected in common bloodwork and confirmed via additional fasting blood tests (LDL-C, lipids, lipoprotein-a) and physical exam. Once diagnosed, the decision of which lipid-lowering statin[6],[7] to use does not rely on the any genetic features. Genetic testing only changes treatment when diagnosis is made due to it – and that’s a pretty small number. Typically, FH patients present LDL-C levels exceeding the 90th percentile (hard to miss). Genetic tests are most valuable for high-risk children, who otherwise might not have their LDL-levels scrutinized. It is little surprise that payers usually restrict coverage of these tests to members with a known first degree relative with the mutation.[8]
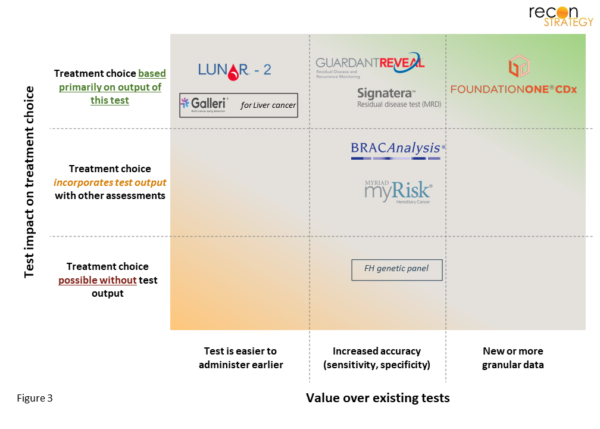
Recently launched tests have focused on immediately-actionable information.
Reviewing how established tests fit into our framework above, it becomes clear that test coverage is easiest when existing evaluations offer little guidance on how to treat. A new test should provide definitive guidance on next-steps; if uncertainty remains (e.g., if a follow-up test is needed), the path is less certain. A few observations and perspectives on coverage status for recently launched tests are below (Figure 3).
Tests to detect residual (or recurrent) cancer cells can be a powerful tool to determine if continued treatment is needed.
Some patients see cancers recur even after being treated with therapeutic-cocktails optimized for their cancer’s traits; adjuvant-treatment (additional time on cancer-drugs) can increase survival, but also lowers quality of life. Minimal Residual Disease (MRD)[9] tests are more sensitive than imaging or standard blood tests that doctors previously relied on to determine if adjuvant-therapy was needed. It is not surprising that Natera has obtained positive CMS coverage for Signatera®[10] and that analysts expect similar coverage for GuardantReveal®. Any tests that similarly rapidly detects cancer recurrence (or failure to eradicate) are likely to enjoy broad coverage.
Early cancer-detection tests must outperform existing screening tools (either in accuracy, ease-of-use, or both).
Present-day screening tests are highly accurate and widely used for breast and cervical cancers (mammograms and PAP smears, respectively). However, for colorectal and lung cancers, there is an opportunity to improve on the SOC screening tool (both colonoscopies and low-dose CT suffer from low adherence).[11] Guardant’s LUNAR-2® for colorectal cancer has similar accuracy, yet is less invasive than a colonoscopy.[12] If studies show the test increases early-stage CRC diagnosis, without increases in false-positives,[13] strong coverage is likely. Less convincing is Grail’s Galleri®, which screens > 50 cancers at once, but has vastly inferior sensitivity compared to SOC screening tests for many early-stage cancers (e.g., in stage I, <20% sensitive for lung cancer, ~50% for CRC).[14] That said, Galleri® could become a primary tool for those cancers that are difficult to detect today, where its sensitivity is strong (e.g., sensitivity >60% for both stage I liver and pancreatic cancers).
PRS test developers should consider whether their test will influence care choices and if changes in patient outcomes are measurable.
Recent gains in genome processing and computational power have made it possible to explore the impact of multiple genes – looking at both deleterious and protective genes, and layering on any interplay amongst them. The approach (termed “Polygenic Risk Scores”, PRS) identifies which patients could benefit from preventative care. In a value-based mindset, what matters is that the test leads to better outcomes: the disease is prevented, delayed, or severity lowered. Diseases where preventative measures are strongly correlated with outcomes have a lower hurdle than those where available clinical actions are limited or unproven. With that in mind, below are a few comments on the opportunities and challenges for select diseases:
The value prop is clear for tests that identify which patients benefit from costly, or novel, drugs.
Both cardiovascular disease (CVD) and type-1 diabetes (T1D) highlight how a PRS tool could be valuable. CVD is very common, with myriad therapeutic options (ACEs, ARBs, statins, PCSK9s) and variable patient response. Here, value lies in optimizing the treatment to the individual. Research to date on PRS for CVD has focused on disease onset;[15] a pivot to response to particular mechanisms-of-action (MOAs) would be useful. If the test predicted who would not respond to MOAs of common generic drugs, it could be used to justify high-cost alternatives (e.g., Entresto, a PCSK9-inhibitor). For T1D, a PRS test that better predicts onset than family history could be a powerful tool for drug development (reducing cohort size for clinical trials of onset-delay drugs, such as teplizumab[16]) and eventually as a companion diagnostic.
When available choices are only loosely tied to outcomes, developers must prove that known “high-PRS” patients fare better than those without PRS testing.
Progress in lung cancer is hampered by patient behaviors (long-term smoking) and low adherence to current screening guidelines.[17] A lung cancer PRS would be justified if individuals with “high-PRS” were proven more likely to quit or get screened than peers without any PRS testing. Proof of these outcomes could require large, real-world studies. Alternatively, for geographies with rapidly rising lung cancer rates amongst non-smokers (e.g., China), a test that stratified non-smokers for increased screening could also be highly valuable.[18] Similarly, for type-2 diabetes, a PRS-test would be most valuable if proven to affect the rates of T2D development, progression to insulin use and emergence of comorbidities.
Concluding thoughts
Improving clinical outcomes, while managing the total cost of care, is paramount for payers. As developers choose which conditions to study, they should consider how they will prove impact to outcomes. The burden of proof will vary, depending on what treatments are available, the timeline of their effect, and how much is known about their efficacy. Below, we highlight key considerations across scenarios:
- When treatments are strongly tied to near-term outcomes (e.g., Keytruda for PD-L1 positive cancers), proof that the test improves selection of the appropriate treatment should be sufficient.
- In cases where treatments are only loosely tied to outcomes (e.g., exercise or dietary counseling), developers should expect more involved studies, showing impact on disease-outcomes.
- If disease-onset itself is uncertain (i.e., tests that stratify patients by disease-risk), evidence the test can lower disease burden (onset rate/timelines, average acuity) will be needed, potentially via real-world studies that control for patient behaviors, environment, and circumstantial-events.
- Biopharma companies could be both early customers (during drug-development) and long-term advocates (after drug launch) for tests that predict patient response to specific therapies.
Sarah Dolman, PhD
Senior Advisor