PDF: Trends in oncolytic virus therapeutics: then and now
The idea of using pathogens to treat disease is not new; ranging from phage therapy to malariotherapy (resulting in the only Nobel in medicine to a psychiatrist – in 1927), but it was really only with the onset of the biotech revolution in the 1990s that an old concept of using viruses as tumor killing agents started to come into its own. As the ability to characterize viruses at the genetic level grew, it was followed by the techniques to genetically modify them to specifically target tumors, increase their cell killing potency and immunostimulatory potential. Thus a new therapeutic modality was born: oncolytic viruses (OV).
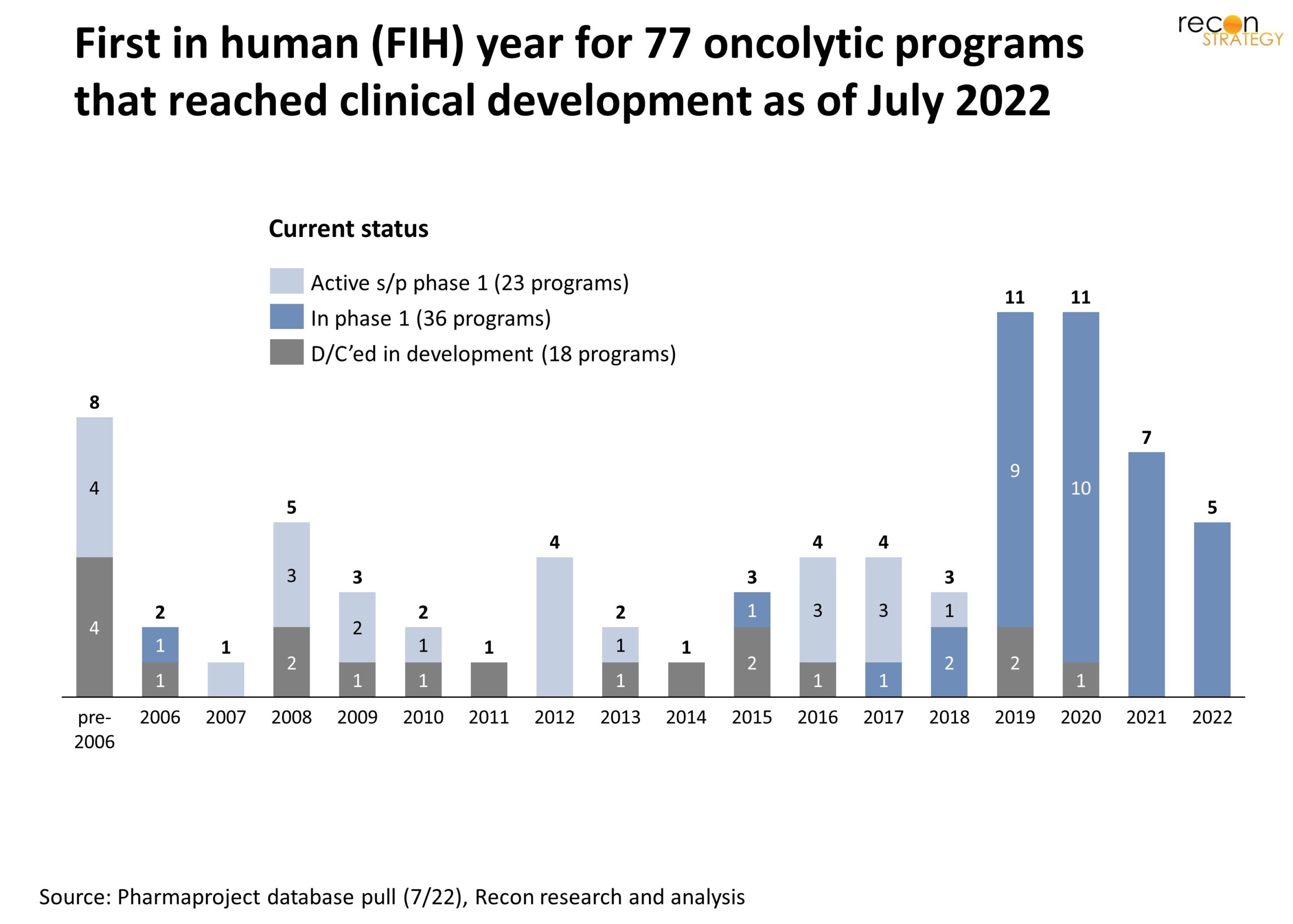
There are a number of excellent reviews[1] of OV therapeutics, but they tend to be descriptions of the current state of the field, rather than capturing how the field has evolved over time, which in and of itself can provide a helpful perspective. To fill this gap, we have sought out and reviewed all oncolytic virus programs that have at least reached clinical development to date (July 2022), including those for which development has since been discontinued. Seventy-seven OV programs have made it into human testing, with 4 reaching approval, though only one in the United States (Amgen’s T-VEC/Imlygic approved by FDA in 2015). The level of activity in this space has been roughly steady from 2006 to 2018 with a significant upsurge of programs reaching the clinic from 2019 onward (Exhibit 1), coinciding with growing inflows of capital in early-stage biotech in general, and cancer R&D in particular.
Exhibit 1
Trends in the biological design of oncolytic virus therapeutics
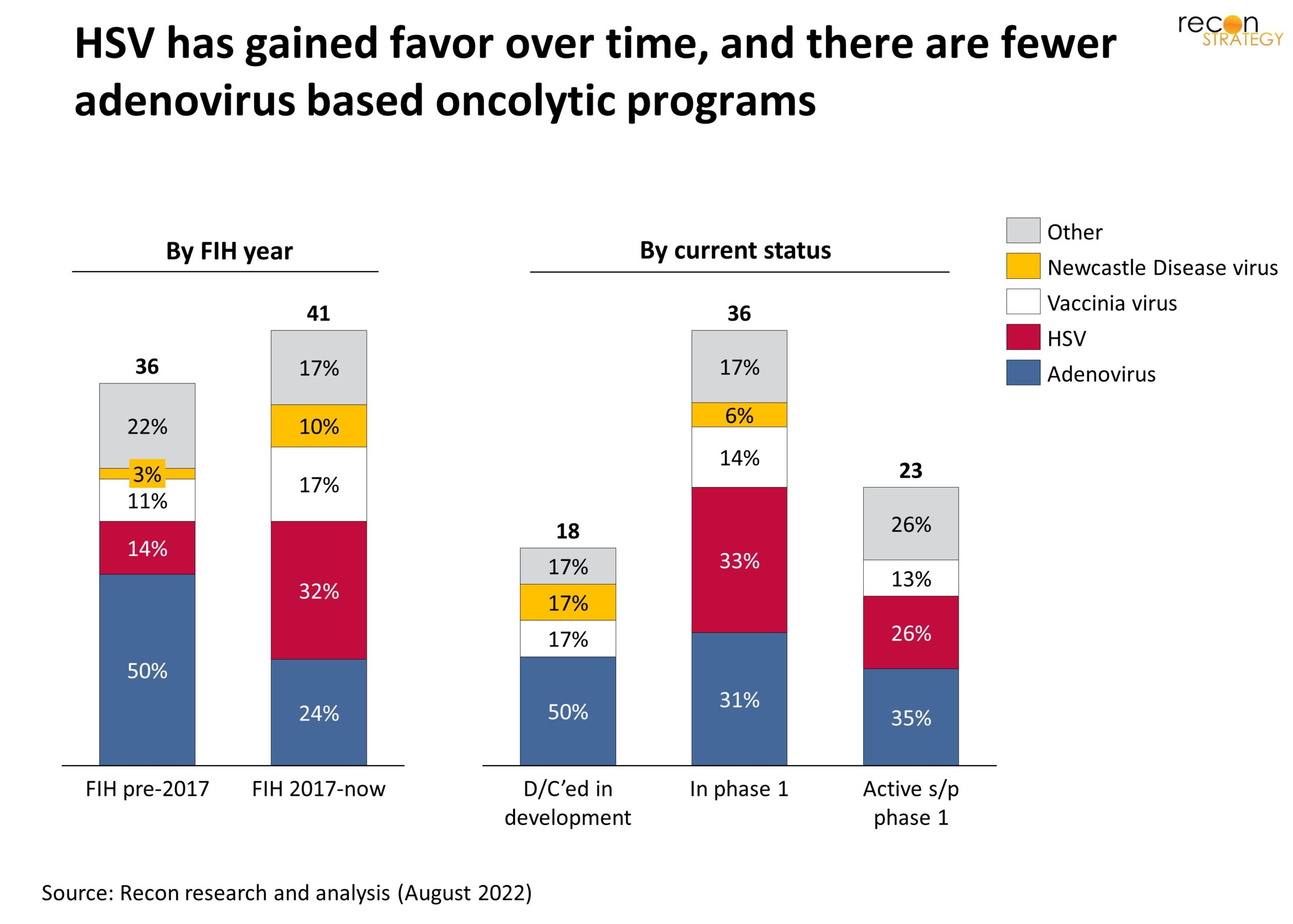
Probably the foremost characteristic of an OV program is the choice of viral platform. While there has been a remarkable diversity of viruses used over time including DNA and RNA viruses (e.g. coxsackie, polyoma, orthopox, etc.), an early favorite has been adenovirus, in part because it has been well studied and characterized and techniques to genetically engineer variants are established. It also has relatively attractive tumor targeting and immunity inducing properties.[2]
However, more recently, herpes simplex virus (HSV) has gained favor relative to adenovirus (Exhibit 2). A factor is that at 154 kilobase, HSV offers a generous capacity for alternative gene payloads relative to adenovirus (35 kb). Although it may be accidental (or reflect that some dormant programs have yet to be terminated), it is notable that few to none HSV OV programs that have reached FIH have been officially discontinued to date. In addition, T-VEC, the only approved OV therapeutic in the US, is HSV-based.
Exhibit 2
In the early days of the field, the main goal of OV therapy was to achieve bulk tumor killing through invasion and destruction of cancer cells by the virus. As a result, back then, a main concern was how to shield the OV from immune clearance long enough to do its work. This perspective has completely changed with the success of immuno oncology approaches. Today, the expectation is that viral infection will guide and boost an immune reaction to the tumor, through the display or release of viral or tumor antigens, and/or induction of immune signaling by infected cells. In that mechanism of action, most of the killing is done by the immune system.
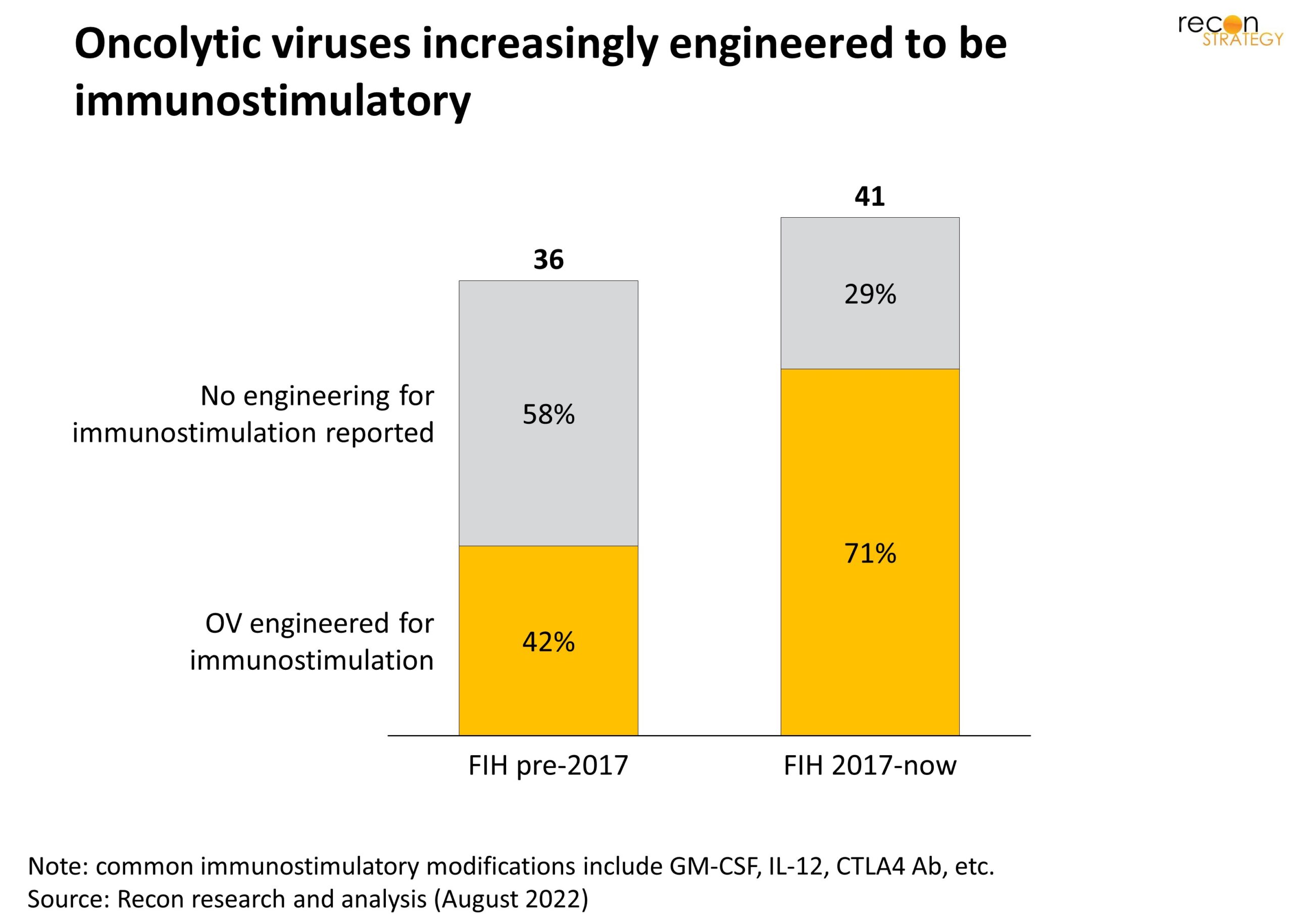
Consistent with that shift, OVs are increasingly engineered to be “armed” with genes encoding immune activating factors which are then transcribed and translated by the infected tumor cells (Exhibit 3). The range and combination of factors that have been implemented in various platforms are remarkable but some cytokines reoccur with significant frequency including GM-CSF (included in T-VEC), IL-12, IL-2. Nor are the factors limited to cytokines, for example a number of OVs encode an antibody against CTLA-4 thus replicating at a very local tumor level the activity of the systemic I/O agent ipilimumab (Yervoy, BMS).
Exhibit 3
Trends in early clinical development strategy
We have systematically reviewed first in human (FIH) studies to highlight how approaches to early clinical development have evolved over time.
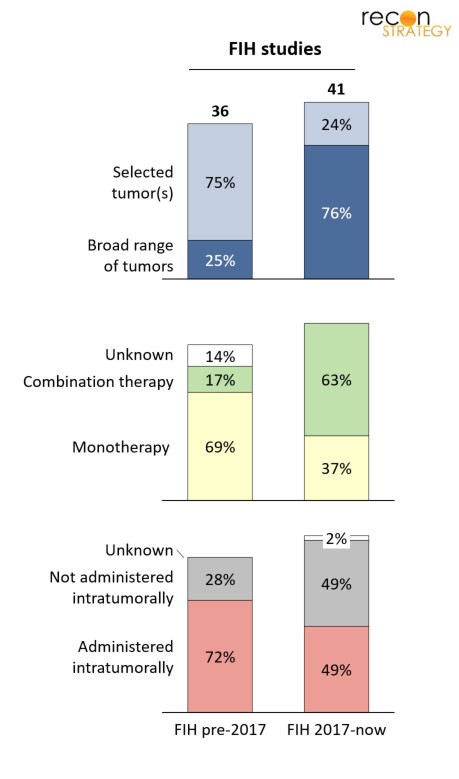
What we found is that older programs that reached FIH before 2017 have tended to zoom in on selected tumors types (with skin – typically melanoma, and brain – typically GBM, most common) in part because of a focus on virus-mediated cell-killing enabled by specific viral tropism toward a given tumor tissue. As immune-stimulation has come to be seen as a dominant anti-tumor mechanism of OV therapy, potential for broader applicability across tumor types has relaxed constraints on initial studies to look for an early efficacy signal in a broader set of cancers. As a result there has been a shift from a minority to a majority of FIH studies accepting patients with a wide variety of solid tumors (Exhibit 4).
Relatedly, there has been a trend toward combination therapy, with pembrolizumab as a frequent companion therapy, further emphasizing the extent to which developers are looking to an immune-driven anti-tumor effect.
Finally, the proportion of initial studies that require intratumoral administration of the OV has been decreasing – and because intratumoral injection can be a constraining factor (the tumor or its metastases must be in a location that is accessible), in many cases this will broaden patient eligibility.
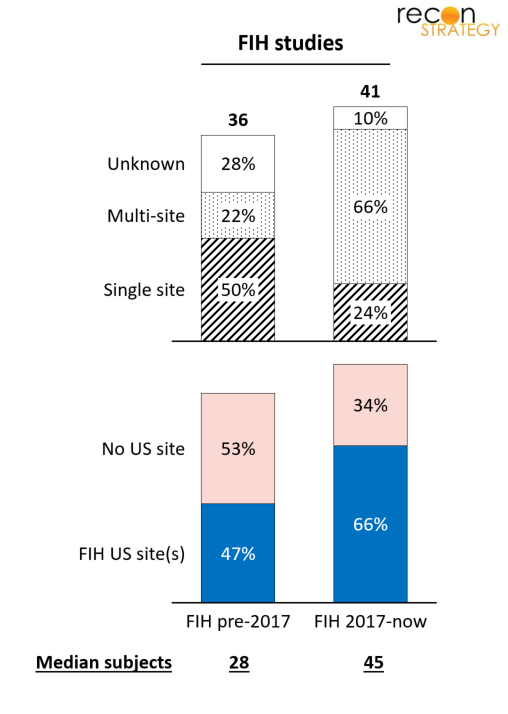
Looking at the operationalization of first in human studies, we found that phase 1 trials are getting larger (pre-2017 median enrollment of 28 vs. 45 from 2017 on) and now usually involve multiple sites (Exhibit 5). This is aligned with a broader trend in oncology in which phase 1 studies are getting bigger, though the paradigm of adding multiple expansion cohorts that lead to triple (and sometime even quadruple) digit enrollment in phase 1 is not yet prevalent for OVs.
Lastly, the proportion of programs with US sites for their FIH trials has been increasing, though international activity, particularly in China remains significant. Whether this reflects the low barriers to capital in the US, regulatory positioning, competitive concerns, the need for a high-level of clinical sophistication, or a mix of all these factors is unclear.
Final thoughts
Even relative to other new therapeutic modalities such as RNAi or gene therapy, the track record of oncolytic viruses is much more limited. There are simply not enough data points to begin to calculate transition rates and probabilities of success. However, looking at 77 programs with first in human experience, it is possible to see substantial changes that have occurred over time in the approach to OVs, particularly the increased use of an HSV backbone and the shift in focus toward an immunostimulatory mechanism of action. Unless there is a therapeutic breakthrough with a program achieving a very high level of success, we estimate that it will take roughly a 2x increase in the clinical experience base to see if these trends are truly making a difference in outcomes. At the current rate of inception of new programs, that’ll be in 5-6 years.
[1] Hemminki, O., Dos Santos, J. M., & Hemminki, A. (2020). Oncolytic viruses for cancer immunotherapy. Journal of hematology & oncology, 13, 1-15; Macedo, N.,… & Kaufman, H. L. (2020). Clinical landscape of oncolytic virus research in 2020. Journal for immunotherapy of cancer, 8(2); Rahman, M. M., & McFadden, G. (2021). Oncolytic viruses: newest frontier for cancer immunotherapy. Cancers, 13, 5452.
[2] Cervera-Carrascon, V., … & Hemminki, A. (2020). Comparison of clinically relevant oncolytic virus platforms for enhancing T cell therapy of solid tumors. Molecular Therapy-Oncolytics, 17, 47-60.