Hematopoietic stem cell (HSC) gene therapy for hemophilia
Roctavian and Hemgenix are in-vivo gene therapies where an AAV vector delivers a functional copy of the defective gene to hepatocytes, the physiological producers of Factor VIII and XI respectively. Issues with those therapies has been their durability (the effect appears to wane over time) as well the fact that pre-existing AAV antibodies preclude their use. But here, an alternative approach is proposed that is analog to CAR-T therapy: HSCs are harvested from the patient, modified with a working copy of Factor VIII, and reintroduced to the patient (after myeloablative conditioning regimen). For the first 5 patients who received this procedure, 3 had quasi normalized Factor VIII activity but all 5 had a zero bleeding rate (prior was 20 events per year or more) that was sustained through the follow-up period (9 to 27 months). A tantalizing result which potentially opens the door to many opportunities, but also raises many questions. Will the response be sustained over a longer time horizon? What is the risk of hematological malignancy? Is it possible to scale back the conditioning regimen (as it is average neutropenic period was 10 days, which is no joke)? And could this be used for other enzyme replacement therapies? Interestingly, this is a US/India collaboration, and the study was conducted in India. Lentiviral Gene Therapy with CD34+ Hematopoietic Cells for Hemophilia A
Anticoagulation: between Scylla and Charybdis
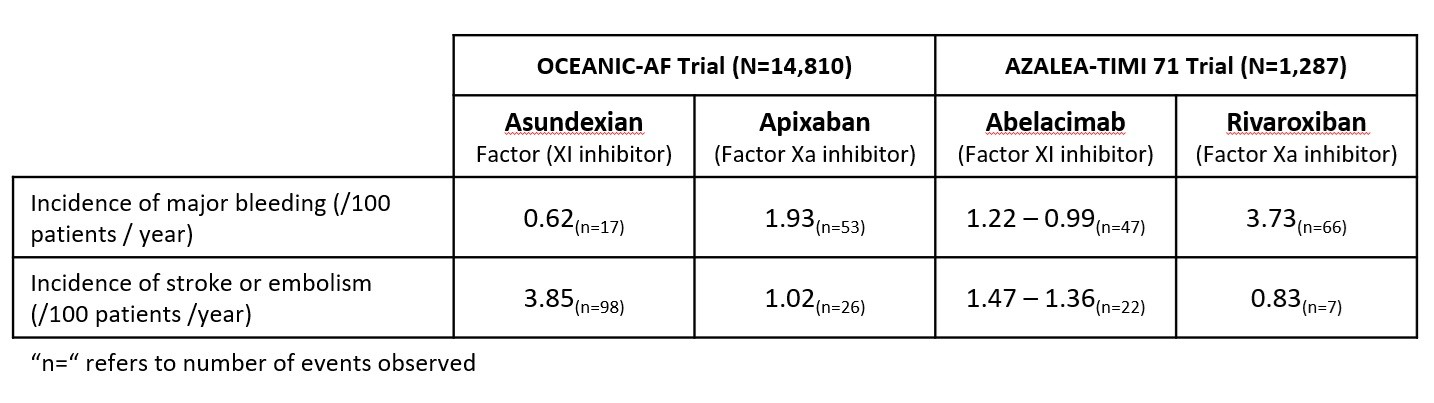
Evolution has finely tuned our coagulation pathways to save us from bleeding to death, but also make sure that we don’t clog our blood vessels with clots. This balance works well for the average healthy person, but for someone with a predisposition toward making clots (e.g. people with atrial fibrillation), blood thinning agents prevent strokes, but at the cost of an increased risk of bleeding. For the past decade Factor Xa inhibitors (rivaroxaban/Xarelto and apixaban/Eliquis) have dominated this space, but more recently, there has been interest in inhibition of Factor XI with both small molecule (asundexian/Bayer) and biologic (abelacimab/Anthos) agents mostly because they have been hypothesized be less prone to lead to bleeding. Both were run in double-blinded RCTs with Factor Xa comparators, and the results are in the table below.
The OCEANIC study comparing asundexian to apixaban was halted by the independent data monitoring committee for an excess of strokes and embolisms vs. comparator, by a factor of about 3x. On the other hand, the incidence of major bleeding was lower by a factor of about 3x. The AZALEA trial was much smaller and testing two dosing regimens of abelacimab vs rivaroxiban. Once again, the bleeding rate was much smaller (by about a factor of 3). But while the trial was not powered to assess impact on stroke / embolism incidence, it does appear that the stroke rate was higher with abelacimab than rivaroxaban though the number of events was small. Is it possible to improve preventative anticoagulation and the trade-off between embolic and bleeding risk? Count me skeptical, at least for a one-size fits all approach. But it may be possible to personalize and optimize treatment at the patient level. Asundexian versus Apixaban in Patients with Atrial Fibrillation; Abelacimab versus Rivaroxaban in Patients with Atrial Fibrillation
A big win in pulmonary arterial hypertension (PAH)
A few years ago, I highlighted a promising new agent for pulmonary arterial hypertension with a different mechanism of action focused on improving the tissue architecture/remodeling of the pulmonary vasculature rather just vasodilation. The originator (Acceleron) was soon after acquired by Merck who has driven sotatercept (Winrevair) to approval in 2024 on the basis of the standard 6 min walk test, a traditional but imperfect measure of efficacy. Now come the results of a longer placebo-controlled study with hard end-points of death, transplantation, or hospitalization that was halted early for benefit (~75% decrease of events in treated patients vs. placebo). Most impressive is that this came on top of patients being on aggressive background treatment (2-3 therapies). Now the next question is whether this therapy will help pulmonary hypertension (PH) related to left heart failure (aka WHO class 2 PH), a big unmet need. We should know more when the CADENCE trial reads out, perhaps later this year. Sotatercept in Patients with Pulmonary Arterial Hypertension at High Risk for Death
A bird flu update
A report on 46 US cases of confirmed, symptomatic, A(H5N1) virus infections in humans (3/2024 to 10/2024). Except for one case, all could be traced to poultry or dairy cow exposures (in roughly equal proportion). Nobody was hospitalized and nobody died – the main symptom was conjunctivitis (“pink eye”). The lack of virulence (and human-to-human transmission) is reassuring although a bit mysterious since death rates in Asia during the mid-2000s outbreak were much higher. Fingers crossed that it stays that way, but it is probably would be wise to be prepared to gear up an mRNA vaccine just in case. Meanwhile, the main consequence of the epidemic seems to be the high price of eggs. Highly Pathogenic Avian Influenza A(H5N1) Virus Infections in Humans
23andyou
A Perspective article on the then impending bankruptcy of 23andme (now well covered in mainstream media). Biggest take-away is that in the context of consumer services, genetic information is not protected health data, and privacy is governed by contract (kind of like the seldom read 50-page EULA one encounters from software vendors). Bankruptcy, Genetic Information, and Privacy — Selling Personal Information
The New England Journal of Medicine is a premier weekly medical journal covering many topics of interest to the health sector. In this series we offer an opinionated perspective on selected highlights that might be of interest to our clients and others.