A couple of years ago, we addressed the question of whether drug companies could use new business models to capture more of the value they create. At the time, we pointed out that drug makers had struggled to get payers interested in new models, and that any potential solution would need to consider aspects of the drug (as it relates to the overall care paradigm and system), and of the payer.
Fast forward to 2016, and there are a number of factors that suggest that now may be the right time for drug makers and payers to partner in creating value by sharing clinical and financial risk. Note the distinction with simple value-based pricing, which you can do prospectively without any risk sharing. While in both cases payers and biopharma companies agree to tie payment for a drug to clinical and/or economic value, rather than quantity of the drug supplied, in a risk sharing arrangement, such payments (or retrospective adjustments to initial payments) are made based on observed performance of the drug relative to promised performance.
There are 3 necessary conditions for risk-sharing to work. These are:
- Uncertainty: (or misalignment) on the value of a drug to patients and/or physicians;
- Win-win: a value proposition that enables both the manufacturer and the payer to see some upside
- Arbiter of value: A viable approach to collect data, determine outcomes, and attribute value
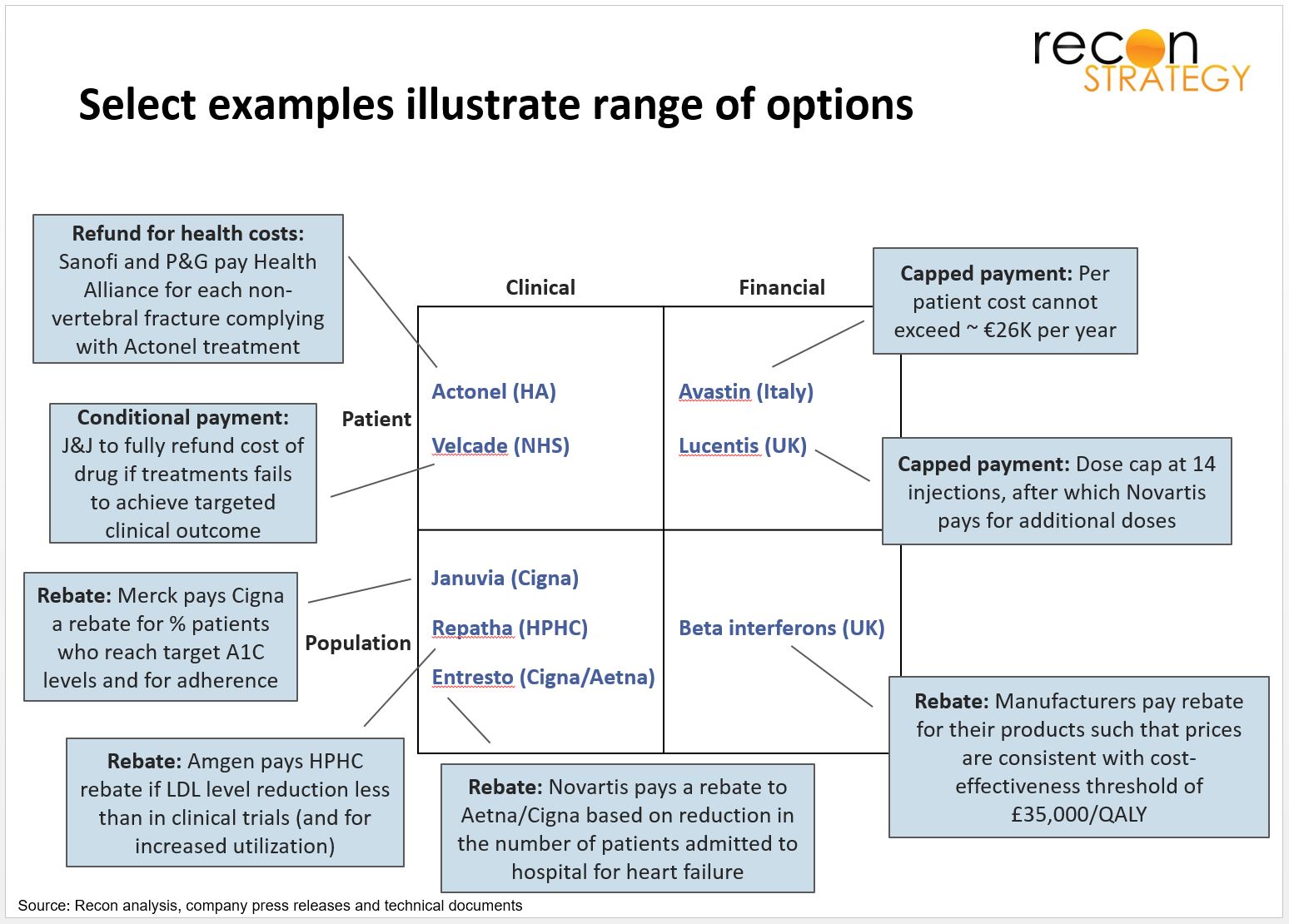
While each situation is different based on factors such as the treatment paradigm and alternatives, downstream costs, and patient/ physician behavior, the most important strategic decision is the type of guarantee to offer and what the relevant unit of analysis should be. Guarantees may be based on various types of clinical outcomes or on economic considerations. The examples below highlight (at a very high level) the broad range of options and some forms of financial recourse.
A core element of risk-sharing strategy is the source of data and metrics. We’ve seen deals that use data that’s from pharmacy or medical claims, patient reported information, purpose built registries, and even a custom study. Increasing interoperability in medical records should result in EHRs becoming more useful especially as the metrics used will increasingly be clinical rather than just financial.
While we’re seeing more interest in these kinds of deals from the biopharma side, they will only succeed in getting payer buy-in if they can genuinely align their interests with those of payers AND can come up with a pragmatic approach. It’s not easy, but it’s doable.