Summary
- Cheap home devices are starting to generate a flood of high frequency, low latency biometric data, much of it of uncertain clinical value
- This uncertainty makes designing the service model difficult: high value use cases may get bundled with broader, low value, more speculative ones (e.g. behavior change), reducing overall ROI and uptake
- Given the patient-generated nature of the data and uncertain accuracy / calibration of the devices, use cases will need specific targeting or depend on subsequent clinical grade investigation to sort signal from noise
- High value use cases are likely going to require tightly designed delivery models to meet ROI hurdles; scaled care management incumbents (rather than technology players or early stage coaching plays) may be best positioned
- As use cases become crystallized, business models of some technology-based entrants — premised on broad data capture / coaching — will see their value propositions de-averaged and high value use cases competed away.
* * *
Low-cost, wirelessly connected diagnostics will generate a flood of high frequency, low-latency patient–developed biometric data. Despite widespread “imagine a world where…” enthusiasm and “spaghetti on the wall” research strategies, the clinical value of much of this data is unclear. Technology is making biometric data much cheaper to generate but the cost of converting it into actionable information remains high and arises in the subsequent investigations needed to sort out true from false positives.
Until there is a clear clinical ROI, the vast majority of providers will have no interest in scouring reams of patient generated data arriving 24/7 for ambiguous signals. Moreover, the vast majority of payers will have no interest in paying the costs for the subsequent investigations to sort ambiguity into clarity.
A clear clinical ROI for high frequency, low latency biometric data needs well-defined, highly targeted use cases which minimize the amount of further investigation required to get to clinical action. The clearer we can be about the use cases, the more effectively can the operating model ensure high clinical value and system acceptance.
Let’s take a potential case in point:
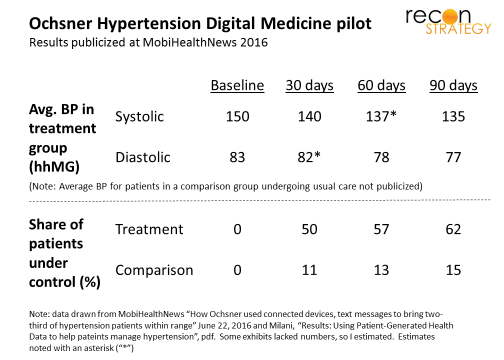
This past June, Ochsner Health publicized results from a pilot using home biometric data collection to manage hypertension (HT) patients. A hundred uncontrolled HT patients used a home blood pressure (BP) cuff to capture and wirelessly share readings with clinicians. These providers engaged patients via text or email with coaching and encouragement. More than 50% of the intervention patients had their HT under control within 30 days vs. 11 percent of patients under usual office-centered care (see exhibit)
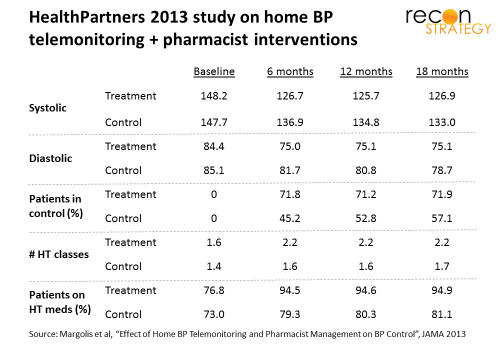
A similar approach was used by HealthPartners of Minnesota in a clinical trial (Hyperlink reported in JAMA in 2013) coupling home BP telemonitoring with pharmacist-patient telephonic engagement. This approach was tested against a control group managed by the usual office visit-centered model. The Hyperlink authors presented a much richer comparison vs. the control group but at greater time intervals than Ochsner. After six months (the first reported observation), 77% of the treatment patients had their HT under control vs. 45% of patients receiving the usual office-based care (see exhibit).
How were these results achieved? The Hyperlink study showed significant changes in the medications: increasing the average number of HT medication classes for each patients from 1.6 to 2.2 (while usual care only increased from 1.4 to 1.6) and getting 95% of patients on HT medications vs. 79% among the usual care patients. The Ochsner study suggests that most of the improvement under this model happens within the first 30 days . This pattern suggests two implications: first, the medication adjustments — rather than slower-acting lifestyle changes — were key; second, the intervention can be have a pretty short duration (e.g., 30 days) and still achieve results.
Both of these studies have issues. Ochsner seemed to require patients to pay for the cuff , almost certainly creating selection bias. The Hyperlink team had to draw on a pool of almost 15K eligible patients arrive at their panel of 450 participants. There certainly is a risk that the trial evaluated what can be done with a very small patient segment seeking active engagement rather than something more broadly applicable. More validation needed!
But: suppose this hypothesis is validated or a model can be designed to broaden uptake by less engaged patients : such an “accelerated cycling to right medication” opportunity would seem generalizable. There are many common conditions (e.g., HT, diabetes, asthma, CHF) where patients are placed on a medication that may not be exactly right but where simple biometric assessments can quickly determine what adjustments are needed.
What kind of service model is best suited to deliver on this kind of opportunity?
A few hypotheses for consideration:
- Owning the connected devices (e.g. scale, BP cuff, blood sugar meter, spirometer, etc.) is unlikely to provide a sustained advantage. Patient operated devices need to be foolproof and therefore simple. The differentiation value of pushing data securely from a device to a cloud is going down as incumbent device manufacturers are increasingly embedding this capability into their latest models. A device-agnostic delivery model offering interoperability with a wide range of device brands would allow the clinical and payer partners to take advantage of their procurement strategies.
- The clinical intervention arising from patient-generated biometric data is likely to be simple, requiring limited additional clinical evaluation to reach a decision: for example, recommending a dosing adjustment or bringing the patient in for further testing. Accordingly, the initial evaluation of the biometric data can be largely algorithm-driven with human escalation options (e.g. to coach patients on collecting data) and a layer of clinical review delivered by physician extenders or pharmacists. In all likelihood, no ongoing relationship with the patient is needed and deep disease knowledge may not even be required.
- Scale can be an important value driver: Just like a call center, the operations will need a certain volume to support rapid evaluation of incoming data, speed to intervention, multiple languages and licensing and the ability to maintain service levels with lumpy patient on-boarding (as the vendor signs up practices).
- The primary barrier to entry is likely to be the construction of customer relationships, for example: having a channel to market the service to payers and providers, having system interoperability with provider EMRs to allow for easy collaboration on specific patients with minimal upfront investment in interfaces and process alignments, having a business function that can contract in a variety of ways to maximize joint value.
Given these conditions, an established healthcare services players with deep pharmacy and care management capabilities (such as a PBM, at-scale pharmacy, specialty pharmacy provider looking to expand into adjacencies, or classic care management outsourcers such as Magellan) would be best positioned to deliver. These kinds of players would have the scale, broad geographic reach, the pharmacy expertise, payer relationships and credibility with providers to provide such a service.
On the other hand, it is hard to image many early stage companies – often leading with technology and positioning themselves as long-term patient coaches — being able to compete on these dimensions. Indeed, I suspect a lot of the clinical value of early stage device/coaching models may be in delivering precisely this type of acute intervention (assessing how a patient responds to changes in treatment over a concentrated interval). If you extract this use case from their value (e.g. if a PBM provides the service on a low-cost acute basis), however, what’s left? The clinical value of high frequency biometric readings for stable, in control patients ambiguous. Providers will have little interest in high frequency data for a stabilized and in control patient. The sustained value of ongoing coaching for out of control patients (who are on the right meds) is ambiguous: proof points are soft and an ROI across a broad population moderate or short-lived (at best).
This puts some start-ups (such as Livongo) in a vulnerable position. They may be trying to build on-going coaching models when the real clinical lift is contained in punctuated episodes around medication adjustments. In time, such cross-subsidization will be ripe for the competitive attack from at-scale players with payer and provider relationships.
It would be ironic if all the VC money being spent on consumer-based biometrics essentially results in a handful of such high value intervention cases that could resuscitate the vendored care management industry.