Download a PDF of this article here.
Hematologic cancers account for ~10% of annual new cancer diagnoses and continue to have some of the poorest overall outcomes, particularly for older adults.[1] However, the last decade has seen major clinical improvements, led by drugs from two new modalities: cell therapy[2] and bispecifics. Here, we discuss the looming competition between these two drug-classes for hematological cancer indications. With recent approvals, and multiple late-stage assets for the same indication, the success of future launches will depend on how treatment paradigms shift, or do not.
Background and context
Over the past 25 years, monoclonal antibodies (mAbs) have become a vital element of oncology treatment protocols. While traditional mAbs target a single tumor antigen to enable an immune response, bispecific mAbs (bsAbs) are capable of binding to two distinct antigens. BsAbs have been researched for ~40 years, with more than 50 different subtypes explored. Today, the most common (27 of 33 active assets) bsAb for hematological cancers are bispecific T-cell engagers (BiTEs), which target antigens on both cancer and T-cells simultaneously, linking patients’ existing T cells to their cancer. However, after decades of investment, only two BiTEs have been approved by the FDA for hematologic cancer: Blincyto, for B-cell acute lymphoblastic leukemia and more recently Tecvayli, for multiple myeloma. [3] Despite the limited numbers reaching approval to date, over 100 US-based BsAb assets are in active development across oncology indications.
Although only in development since 2012, autologous CAR-T therapies (which reengineer patients’ own T cells to attack cancer) have seen more success, with the first approval in 2017 of Kymriah for B-ALL. Kymriah has a much higher overall remission rate compared to Blincyto (81% vs. 34-66% for pediatric B-ALL patients).[4] However, CAR-T therapy is hindered by a complicated manufacturing process, requiring patients to wait days-to-weeks for their manufactured T-cells. Five other CAR-T therapies have since been approved for hematological cancers, all with similar production complexity. Developers are exploring several means to circumvent the challenges of autologous products, including different cell types, different methods for genetic modification, and allogeneic (non-patient derived) approaches. In total, there are 200 US-based cell therapies in clinical trials – 85 for hematological cancers.
Perhaps driven by the handful of recent successes, the oncology cell therapy pipeline is now more than double the size of the bsAb oncology pipeline. In the past five years, new phase starts in both modalities appear to have similar growth rates (Figure 1). While new starts for cell therapies are led by preclinical programs, bsAbs growth is more heavily weighted in clinical trial starts. At first glance, the preclinical bsAb pipeline appears comparatively small; however, because bsAbs are more often led by large pharma, it is possible many are undisclosed (more below).
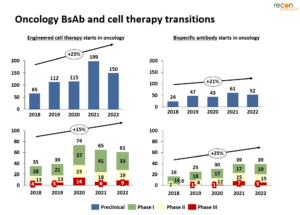
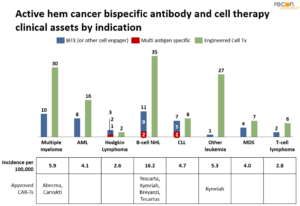
Given the large success of CAR-T therapies, one might expect the bsAb development pipeline in hematological cancer to focus outside of the 3 indications addressed by approved CAR-Ts. While there is a higher proportion of the bsAb pipeline in some indications without CAR-T approvals (CLL, ALL) compared to cell therapies, most BiTE development is in multiple myeloma (MM) and B-cell non-Hodgkin lymphoma (BC-NHL), where the cell therapy pipeline is also concentrated and multiple CAR-Ts are already are approved, per Figure 2.
Multiple characteristics of MM and BC-NHL make these indications attractive for drug developers. Significant need remains for both indications: ~50% of patients with DLBCL (the most common form of BC-NHL) and almost all patients with multiple myeloma become refractory to or relapse after first-line treatment.[5] Later-line treatments also leave room for improvement – approved CAR-T therapies are not universally curative, resulting in a new class of CAR-T refractory/relapsed patients. Further, both MM and BC-NHL have multiple known tumor antigens that do not present widely on normal cells. In contrast, known antigens for other heme-cancers are also largely expressed on healthy cells (e.g., T-cell lymphoma). In the US, five BiTEs targeting these two indications are late-stage (Ph3 or nearing registration) and 17 cell therapies are in phase 1/2 (none beyond).
Will BiTEs break into indications currently served by CAR-T?
Given the high number of programs, it seems likely patients will soon have multiple treatment options, and until an effective cure is found, these products will likely compete for patient and physician preference at each time-point in the treatment-pathway (i.e., 1st-line, 2nd-line, etc.). To understand which assets will be preferred, it is critical to consider: (1) efficacy and safety, (2) drug-access and treatment protocol (3) impact on the entire treatment pathway, and (4) developer resources and investment. We explore each in depth below.
- Efficacy and safety
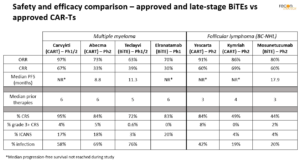
Any assets with superior clinical efficacy and safety data for a particular disease or patient group, will readily displace commercial CAR-T treatments. Most BiTE clinical trials investigate use in relapsed/refractory patients, which are on-label for commercial CAR-Ts. To date, BiTEs’ response-efficacy is roughly equivalent, not superior, to CAR-Ts in trials (Figure 3). However, in terms of sustained response and possible cure, CAR-Ts have an advantage – only requiring a single dose, after which engineered cells engraft and continue to proliferate. In contrast, BiTEs have a short half-life, resulting in the need for repeat dosing to maintain response. Safety differences are largely negligible as well. Cytokine release syndrome (CRS) is the most prevalent adverse event seen to date across both modalities, occuring at similar rates in BiTE and CAR-T trials. Rates of infection and ICANS have also been seen at similar rates. However, grade 3+ CRS (which can lead to serious and potentially fatal complications such as HLH/MAS) has appeared more often in CAR-T than BiTE trials. While ramp-up dosing regimens can be used to further reduce adverse events for BiTEs, there are approaches that could reduce the risk for CAR-Ts as well. While further behind ramp-up, embedded suicide or on/off switches could substantially reduce the rate of severe AEs in CAR-Ts.
While developers are beginning to evaluate assets from both modalities as first-line therapy (e.g., Yescarta, glofitamab, mosunetuzumab are being studied in treatment-naive[7]), it remains too early to know if either will have an edge in safety or efficacy. If any of these therapies are found to have high-efficacy as first-line treatment, along with long durability, the pool of R/R patients for later-line treatments will naturally decline.
- Drug-accessibility and impact on end-to-end treatment
Even in the absence of a clear efficacy or safety advantage, BiTEs may yet serve some patients – particularly since CAR-T treatment access is limited to specialized centers and requires arduous preparative treatment (including apheresis and chemo-preconditioning). Frustratingly, even once a patient’s cells have been successfully collected, CAR-T manufacture is lengthy (~3 weeks vein-to-vein, on average) and not guaranteed (e.g., manufacturing fails for ~10-15% of ALL patients). In contrast, BiTEs could be broadly distributed (well beyond today’s CAR-T centers) and would not require pre-treatment cell-collection or conditioning. Therefore, BiTEs might serve several patient segments, including:
- Patients unable (or unwilling) to travel or wait for an open CAR-T slot
- Patients awaiting a slot for CAR-T (pre- or post-apheresis, as a bridging therapy)
- Patients for whom CAR-T manufacture fails
These considerations are only amplified when considering global demand and supply. Today, there are few CAR-T treatment centers, and even fewer commercial manufacturing sites; both of which severely restrict access. Even with the growth of CAR-T-delivering sites in Central and Eastern Europe, and plans to build additional manufacturing facilities[9], long wait-times to get on treatment schedules are likely to remain. Of course, expanding beyond Western markets, will require an inflection in the price-point – something unfeasible with the cost-of-goods for current CAR-T manufacturing.[10]
>
The above considerations will hold, as long as all CAR-T products are autologous. However, as discussed in an earlier blog post[11], many firms are investing in allogeneic cell therapies, which could be as broadly available “off-the-shelf” as BiTEs. While untested, allogeneic therapies could rapidly become a direct competitor for these segments.
- Impact on entire treatment pathway (1st-line to last-line)
Another factor to consider is how the addition of each new therapy affects the entire treatment pathway – i.e., the preferred order and choice of therapies. Preliminary evidence suggests both BiTEs and CAR-Ts can lead to antigen loss, which could hinder later use of therapies that target the same antigen.[12] The recently approved Tecvayli as well as other BiTEs in development for MM risk this direct conflict, as they target the same antigen (BCMA) as approved CAR-Ts. Interestingly, Janssen is investigating Tecvayli as an early-line treatment despite the risk it will reduce the r/r MM population treated by their commercial CAR-T, Carvykti.[13]
In BC-NHL, all late-stage BiTEs target a different antigen than commercial CAR-Ts (CD20 vs CD19, respectively), so they could be used in sequence. However, the cell therapy pipeline includes several therapies designed with dual antigens that target both CD20 and CD19.[14] If this multi-antigen approach grows, the opportunity set for BiTEs will become tighter and tighter for fear of robbing patients of future treatment options.
- Developer resources and investment
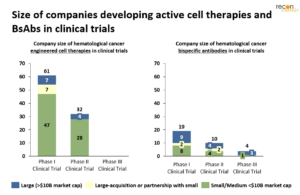
Finally, the resources and funding that drive clinical execution can have an impact on clinical timelines and success. The majority of the cell therapy pipeline is sponsored by small (<$10B market-cap) biotech companies, while most BiTE developers are large (>$10B market-cap) pharma (Figure 4). The influx of money into early biotech in between 2017 to 2021 enabled many preclinical cell and gene therapy firms to dive into clinical studies, rather than license out after pre-clinical development. However, with the recent market downturn, causing over one-third of biotechs to lose 75% of their value since early 2021,[15] it is unclear how the bulk of the cell therapy pipeline will move forward. While large pharma may buy many, there is a question of which assets will be abandoned, opening up the space for BiTEs and other modalities.
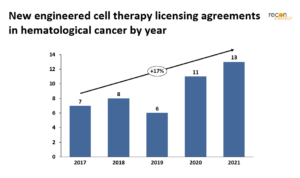
The past year has seen a number of acquisitions of cell and gene therapy firms (or assets) by large pharma. The majority of these acquisitions seem to be driven by in vivo gene therapies for rare disease (e.g., AstraZeneca acquisition of LogicBio, Novartis’ scoop up of two ophthalmological gene therapy biotechs[16]) or cell therapies for solid tumors (e.g., Takeda’s acquisition of GammaDelta Therapeutics[17]). While licensing agreements for cell therapies targeting hematological cancers (Figure 5) has grown steadily, the rate tracks the overall cell therapy pipeline – it has not spiked or altered with the recent biotech industry downturn.
Concluding thoughts
Today, BiTE and CAR-T hematological development pipeline largely overlap – both focus on r/r MM or BC-NHL patients. While new BiTEs will inherently have less real-world safety data, they could address unmet needs for multiple patient segments: (1) patients for whom CAR-T has failed (relapse post-CAR-T treatment) (2) patients needing bridge-treatment while awaiting CAR-T, or (3) patients unable to access CAR-T (due to ineligibility or manufacturing failures). Use of BiTEs even within these segments could change going forward with advancement of improved cell therapies that fill these gaps in treatment. However, whether the cell therapy pipeline can continue its rapid advance remains to be seen in the current funding environment. While many small biotechs are currently struggling to find funding, large-pharma might find these assets compeling at current valuations. Approval of allogeneic cell therapies or developments in CAR-T manufacturing would diminish the need for BiTEs in these circumstances, but new innovations may also increase addressable segments through the use of these assets in tandem (i.e., combo BiTE-CART therapies) – preclinical studies have shown promise.[19]
Ultimately, the key criteria for the success of BiTEs and CAR-Ts as individual therapies will be efficacy and safety – which asset has the best outcomes, or even offers a cure. In the absence of a curative option, the choice between a BiTEs or a CAR-T that targets the same antigen will come down to whichever one gives the greatest chance for long-lasting remission. New therapies that target unique antigens may be used in addition to approved therapies, and therefore the “gold standard” treatment regimen will likely continue to evolve with the best CAR-T or BiTE until a curative therapy materializes.
Maggie Pickard
Associate Consultant
Sarah Dolman, PhD
Partner
[1] (1) Cancer Facts & Figures 2022, American Cancer Society (ACS), Atlanta, Georgia, 2022, (2) Krok-Schoen, Fisher, et al. Incidence and survival of hematological cancers among adults ages ≥75 years. Cancer Med. 2018
[2] In the context of this paper, we define “cell therapy” as any genetically engineered therapy using cells. We exclude non-modified cell therapies.
[4] (1) Queudeville, Ebinger. Blinatumomab in Pediatric Acute Lymphoblastic Leukemia-From Salvage to First Line Therapy (A Systematic Review). J Clin Med. 2021 (2) Maude, Laetsch, Buechner, et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N Engl J Med. 2018
[5] (1) Crump, Neelapu, Farooq, et al. Outcomes in refractory diffuse large B-cell lymphoma: results from the international SCHOLAR-1 study. Blood. 2017, (2) Rajkumar SV. Multiple myeloma: Every year a new standard? Hematol Oncol. 2019
[6] All indications in clinical trials (Clinicaltrials.gov) for each active US-based asset (via PharmaProjects)
[7] (1) Yescarta ZUMA-12 study, (2) Glofitamab COALITION study expansion, (3) Mosunetuzumab in People With Follicular Lymphoma
[8] (1) Berdeja, Madduri, Usmani, et al.; Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet. 2021, (2) Munshi, Anderson, Shah N, et al.; Idecabtagene Vicleucel in Relapsed and Refractory Multiple Myeloma. N Engl J Med. 2021, (3) Moreau, Garfall, Niels, et al.; Texlistamab in Relapse or Refractory Multiple Myeloma. N Engl J Med 2022, (4) Sebag, Raje, Bahlis,et al.; Elranatamab (PF-06863135), a B-Cell Maturation Antigen (BCMA) Targeted CD3-Engaging Bispecific Molecule, for Patients with Relapsed or Refractory Multiple Myeloma: Results from Magnetismm-1. Blood 2021, (5) Bouchkouj, Zimmerman, Kasamon, et al. FDA Approval Summary: Axicabtagene Ciloleucel for Relapsed or Refractory Follicular Lymphoma. Oncologist. 2022, (6) Fowler, N.H., et al.Tisagenlecleucel in adult relapsed or refractory follicular lymphoma: the phase 2 ELARA trial. Nature Medicine. 2021, (7) Budd, Sehn, Matasar, et al. Safety and efficacy of mosunetuzumab, a bispecific antibody, in patients with relapsed or refractory follicular lymphoma: a single-arm, multicentre, phase 2 study. Lancet Oncol. 2022
[10] Pharmaceutical Technology, Apr 2022
[12] (1) Zhou, Liu, Ren, et al. The landscape of bispecific T cell engager in cancer treatment. Biomark Res 2021, (2) Majzner, Mackall. Tumor Antigen Escape from CAR T-cell Therapy. Cancer Discov. 2018
[13] Teclistamab and Darzalex phase I clinical trial, 2022
[14] Shah, N.N., Johnson, B.D., Schneider, D. et al. Bispecific anti-CD20, anti-CD19 CAR T cells for relapsed B cell malignancies: a phase 1 dose escalation and expansion trial. Nat Med 2020
[15] PharmExec, “Are We Closer Than Ever to a Biotech Bottom?”, Jun 2022
[16] Astrazeneca, Oct 2022, Novartis, Dec 2021 , Novartis, Sept 2021
[18] All licensing agreements, regardless of market cap
[19] (1) Choi, Yu, Castano, et al. CAR-T cells secreting BiTEs circumvent antigen escape without detectable toxicity. Nat Biotechnol, 2019 , (2) Yin, Rodriguez, Li, et al. Locally secreted BiTEs complement CAR T cells by enhancing killing of antigen heterogeneous solid tumors. Mol Ther. 2022
