A PDF version can be downloaded here.
One of the major aims of the proposed BIOSECURE ACT[1] is to reduce supply-chain risks for pharmaceuticals, by penalizing firms that contract with China-based “biotechnology companies of concern” — specifically naming WuXi AppTec and WuXi Biologics. Nearly all discussion in the media has focused on the impact to drug-manufacturing; yet the contribution of these firms to innovation may be even greater. The industry must plan ahead — to future-proof not just supply chains, but also product-pipelines and diversity of CDMO partners. Here, we delve into these businesses, highlighting the magnitude of their involvement in R&D and the development of new drugs.
WuXi AppTec and WuXi Biologics: Major players in drug development
Today, the bulk of WuXi AppTec (WXA) revenue (75%) derives from their manufacturing strength — primarily GMP and non-GMP supply of small molecules, with a smattering of advanced therapeutics (CAR-T, TIL, MSC and associated viral vectors), as highlighted in Figure 1.[2] In contrast, the share from safety testing and clinical-operations is modest (16%), with a limited contribution from drug-discovery (8%). While WuXi Biologics (WXB) does not report a similar BU-breakdown of revenue (instead using program stage),[3] it seems their activities are predominantly focused on drug supply (specific to proteins, peptides, antibodies and antibody-drug-conjugates), with no reference to safety testing or clinical CRO services. Despite no directly outlined revenue due to drug-discovery, work described by WXB includes both target and structure elucidation — surely a part of pre-IND phase programs.
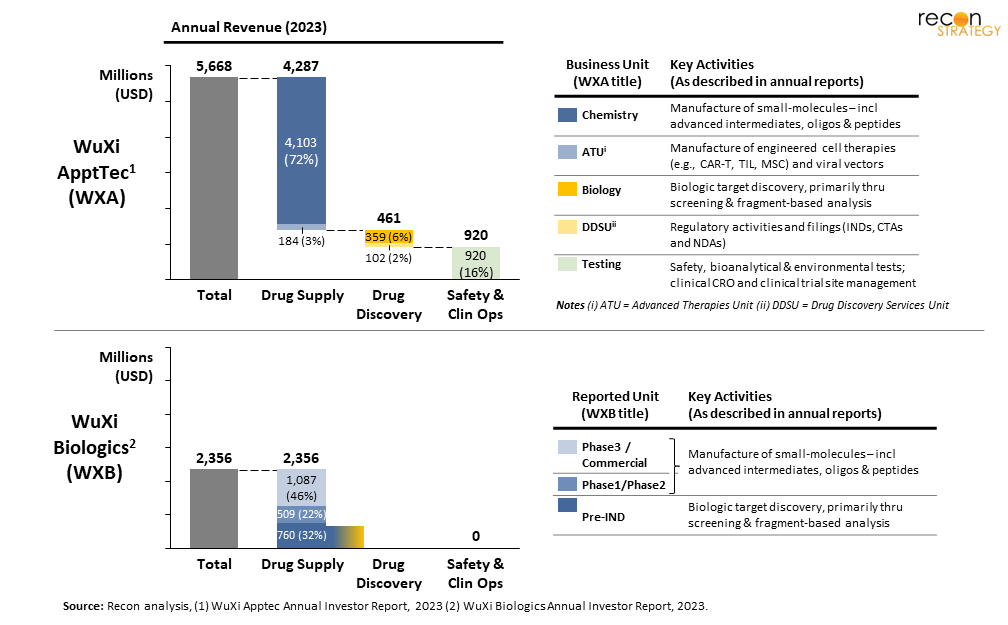
In the general media, most of the discourse on BIOSECURE has focused on the risk to drug manufacturing, and potential for a negative impact on American access to drugs.[4] While the intent of BIOSECURE may be to incentivize US-biopharma / biotech to use alternate vendors (and possibly even in-source, or repatriate manufacturing activities), greater consequences are likely — both WXA and WXB have become increasingly critical to new drug discovery and development. Much of the materials sourced at these firms is critical to drug-development: both drug substance used in animal studies and clinical trials as well as complex chemicals used for rapid drug-discovery. In fact, when we look by program stage, these firms support far more early-stage (preclinical and phase 1) development than late-stage (Figure 2) — a figure that has steadily increased in the last five years. WuXi’s stated strategy of “follow the molecule” has led both firms to seek partnerships with customers at ever-earlier stages of drug development.
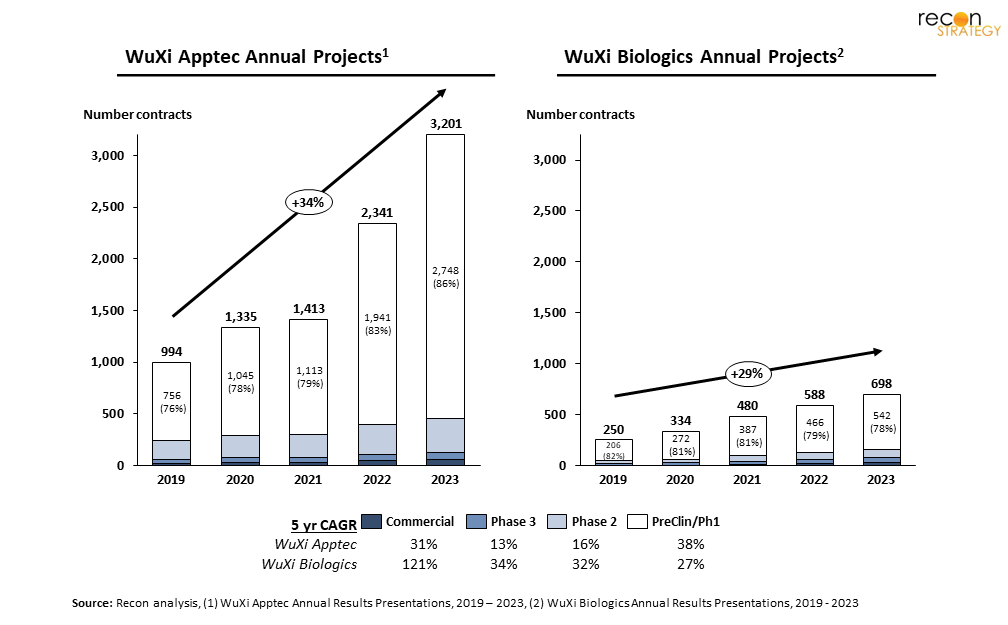
Industry-execs are well aware of the risks: a recent BIO survey[5] of biotech executives and investors found that 75% report a reliance on China-based CDMOs. Of those, 53% expected it would be “extremely difficult” to source viable alternatives, and 64% expected a “substantial slowdown” to their product pipeline, if BIOSECURE were enacted. We spoke with executive R&D leaders at three of biopharma’s top-10; each confirmed they are already working to evaluate alternatives (including for preclinical), and expect some challenges as programs are moved. Recent updates to the bill[1] have created a grace period before enforcement (now set at 2032[6]), yet that is little time for capacity to be built onshore. Separately, the White House has passed several incentives to encourage more US-based capacity; as we discussed last year, the funding offered likely needs substantial matching (or exceeding) investment from industry-players.[7]
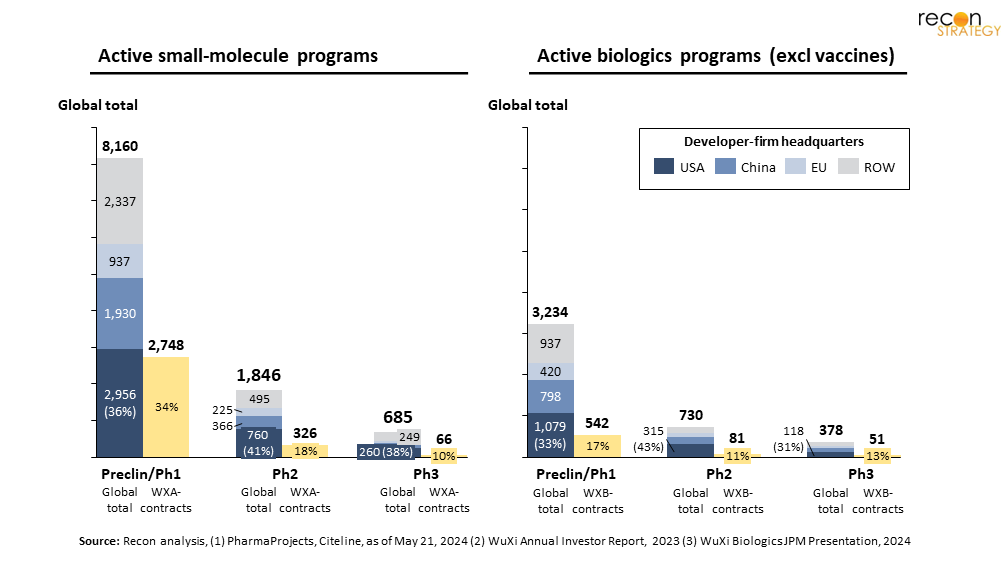
Certainly just about every CDMO seeks to engage early in the drug-development life-cycle — it positions firms better to win the higher-value commercial supply contract. However, the program numbers represented by these firms are non-trivial. Consider how WXA and WXB project numbers compare to the global total of publicly-disclosed programs (Figure 3);[8] these firms have become significant players in early-development, supporting 34% and 17%, (WXA and WXB respectively) of all early-stage programs. Unless there is substantial unutilized capacity at alternate CDMOs, classic economic laws of supply-and-demand suggest a large increase in the cost to progress these programs, with delays likely as well. Small and emerging firms, often the harbingers of the most innovative preclinical assets, are those likely to suffer most. With limited financial resources, these firms often leverage the flexibility of an outsourced model for lower value lab-work. Increasing R&D cost is more likely to reduce the concurrent pursuit of multiple assets by these emerging firms.
Conclusion
Over the last 20 years, the biopharma industry has diversified much of its value-chain — today spanning everything from early R&D to commercial manufacturing. Outsourcing to low-cost geographies has increased capital flexibility for the industry, via a reduced workforce and fewer wholly-owned production sites. The challenge for US-based firms, will now be whether or not they continue to externalize as much of their early-discovery work, and if so, where to source alternative CDMOs.
References
[1] H.R. 7085 – BIOSECURE Act, 118th Congress (2023-2024), Sponsored by Rep Gallagher (R, WI). (a) Original draft introduced Jan 25, 2024; (b) Amended draft introduced May 10, 2024.
[2] WuXi AppTec Annual Report, 2023 (link) and summary presentation (link)
[3] WuXi Biologics Annual Report, 2023 (link) and JPM presentation (link)
[4] (a) Jewett, J. Chinese Company Under Congressional Scrutiny Makes Key U.S. Drugs. The New York Times. April 15, 2024 Accessed May 26, 2024, https://www.nytimes.com/2024/04/15/health/wuxi-us-drugs-congress.html (b) Wainer, D. U.S.-China Decoupling Poses Supply-Chain Risks for Drug Companies. The Wall Street Journal. March 23, 2024. Accessed May 26, 2024. https://www.wsj.com/tech/biotech/u-s-china-decoupling-poses-supply-chain-risks-for-drug-companies-c17e124c
[5] Fishburn, CS. Anti-China bills portend massive blow to biotech: BioCentury survey. BioCentury. March 22, 2024 Accessed May 22, 2024, https://www.biocentury.com/article/651851
[6] Pratap, A. Amended Biosecure Act sets new timeline to move away from Chinese outsourcing firms. c&en. May 15, 2024. Accessed May 27, 2024 https://cen.acs.org/business/outsourcing/Amended-Biosecure-Act-sets-new/102/i15
[7] Our take on some of the recent federal government initiatives aimed at repatriating drug-manufacturing last year: Wiesenthal, J., and Dolman, S. J. Is the future of US pharma manufacturing domestic? Recon Strategy, 2023. https://reconstrategy.com/2023/02/is-the-future-of-us-pharma-manufacturing-domestic/#_edn2
[8] Recon Analysis, PharmaProjects via Informa-Citeline. Accessed May 15, 2024.
